Researchers from the CU School of Medicine Department of Orthopedics have been awarded a $3.4 million National Institutes of Health grant to study the genetics of bone density and to look for therapeutic targets to counter bone loss.
With the grant, Cheryl Ackert-Bicknell, PhD, associate professor of orthopedics at the CU School of Medicine, plans to map the key genes and pathways involved with bone cell activity. Using that information, researchers hope to find targets for more effective treatments to counter bone loss. Ackert-Bicknell’s fellow principal investigator on the grant is Charles Farber, PhD, associate professor of public health sciences at University of Virginia.
Bone is in a constant state of remodeling, Ackert-Bicknell said, and her study is designed to look at cells known as osteoblasts, which work to build bones. When a healthy body is functioning properly, osteoblasts work in balance with other cells called osteoclasts to maintain sufficient bone mineral density.
“In the process of walking from your car to here, you did microcrack damage to your bone,” Ackert-Bicknell explained. “The osteoclasts’ job is to home to that site of the microcrack, eat a hole out all the way around that microcrack and the osteoblast comes and fills that in. That’s called normal bone turnover. That’s most of your life. If the osteoclasts go haywire and there is insufficient building of new bone, that’s osteoporosis.”
Current therapies to cause the body to build the right amount of new bone are limited. These therapies can often only be used for a limited time, and none can be used in children. There is a need for better therapies, Ackert-Bicknell said, but they cannot be developed without improved understanding of how a healthy body gets bone density just right.
That’s where Ackert-Bicknell’s study comes in. She plans a comprehensive analysis of how the genes influence the process. She will conduct a genome-wide association study that seeks to identify genetic variations that are associated with osteoblast function and bone mineral density.
“Osteoblasts build bone. Osteoclasts chew up bone. And that’s how I always teach it: Blasts build and clasts chew,” Ackert-Bicknell said. “Osteoblasts do two things to make bone. They make a protein matrix and then they mineralize that matrix. For that to be accomplished, the osteoblast has to get to the right place, and it has to proliferate. So, it is proliferating, it is migrating, then it is making that bone by making matrix, and it is mineralizing that matrix.”
Previous studies of osteoblasts have shown that its characteristics are highly heritable, or transmissible from parents to children. But how osteoblasts form and do their work is not fully understood. What might appear to be a small change on osteoblast behavior can have significant developmental consequences. Ackert-Bicknell cited an example of how knocking out a single gene in one type of bone cell can result in an obese mouse.
“You can actually knock out a gene, just in these osteoblast bone cells, and get an obese mouse,” she said. “In that cell only. It is the only cell in the whole body that makes that gene, and you end up with an obese mouse. This just shows how bone is tied into all of physiology.”
To get a better understanding of those connections, Ackert-Bicknell’s new study will look at the network of all the genes expressed in this cell and their relative expression in different contexts of genetics.
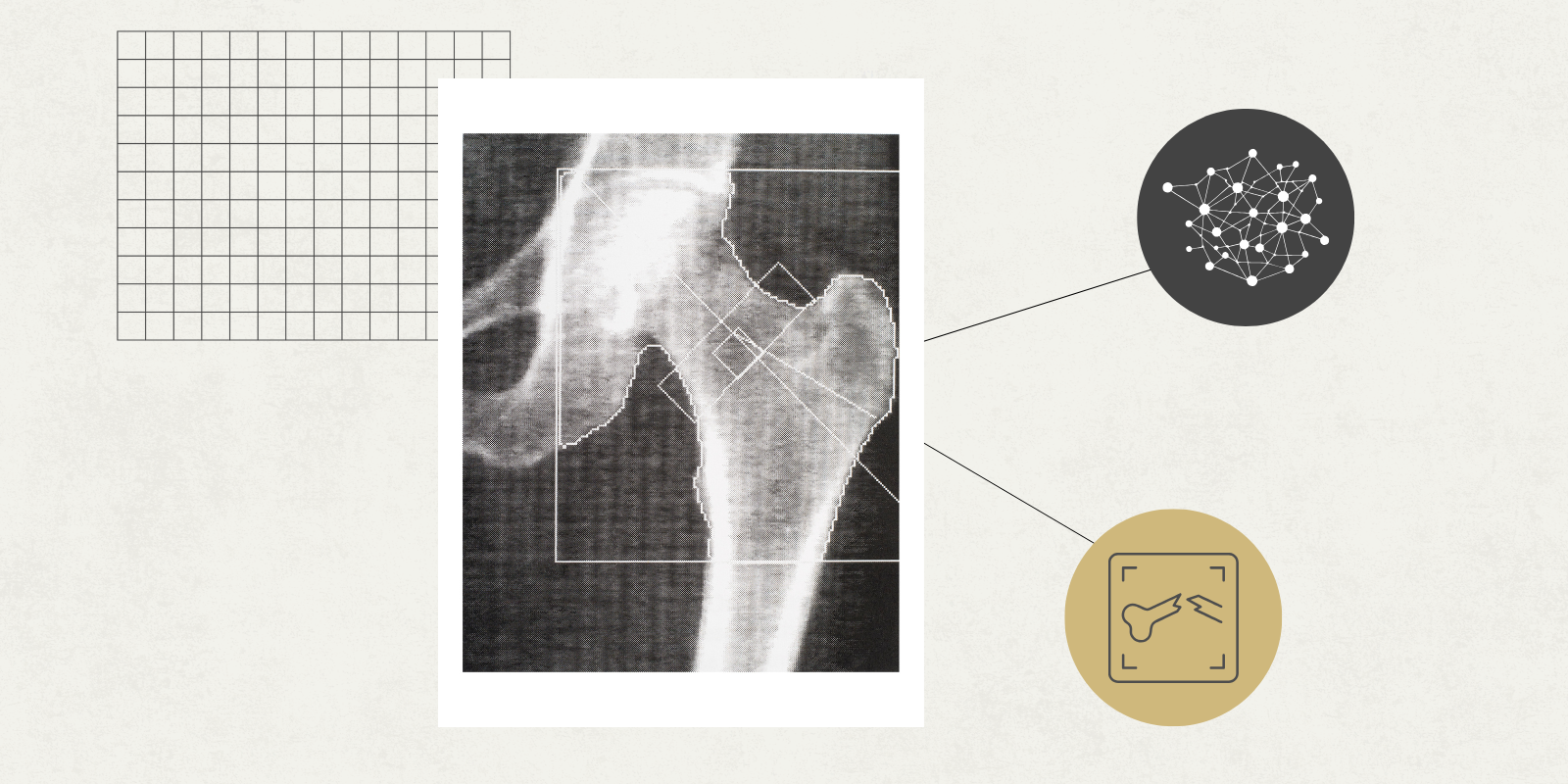
“To do this, we must compare bone image after bone image to identify variations that could be meaningful in relation to the genetics,” said Douglas Adams, PhD, associate professor of orthopedics, who has worked with Ackert-Bicknell on previous studies that underpin the work in this new award.
Adams is also working with Ackert-Bicknell on another NIH grant studying how the leading treatment for osteoporosis might have variable efficacy because of genetic differences between patients. To conduct such studies, researchers grind through thousands of data points to discover links that have yet to be uncovered. By identifying those unknown connections, the team hopes to discover new ways to treat disorders of bone mineralization.
“It’s sort of along the lines of looking under the streetlamp for your keys,” Ackert-Bicknell said. “As long as your keys fell down where the lamp is shining light you’ve got a good chance of finding them. In our work, we are looking outside of the streetlight for the things we haven’t studied before.
“Let’s face it, what we know now isn’t giving us enough drug targets, enough information. It isn’t helping us. The most unique pathways and the ones that are going to get us drugs are not the ones we have already studied.”
Ackert-Bicknell’s new grant provides funding for a five-year research project. Coupled with the parallel active NIH grant held by both Ackert-Bicknell and Adams, CU Orthopedics is at the forefront of the effort nationally and globally to understand and develop disease modifying approaches to address bone loss in osteoporosis patients. These projects are just part of a growing portfolio of research activity in the Department of Orthopedics, which has seen a greater than 10-fold increase in extramural grant support since the department’s leadership committed to expand its research mission in 2018.