AURORA, Colo.– Galaxy Ophthalmics and CU have completed an exclusive option agreement to allow the company to commercialize an implantable medical device to help prevent loss of vision resulting from glaucoma.
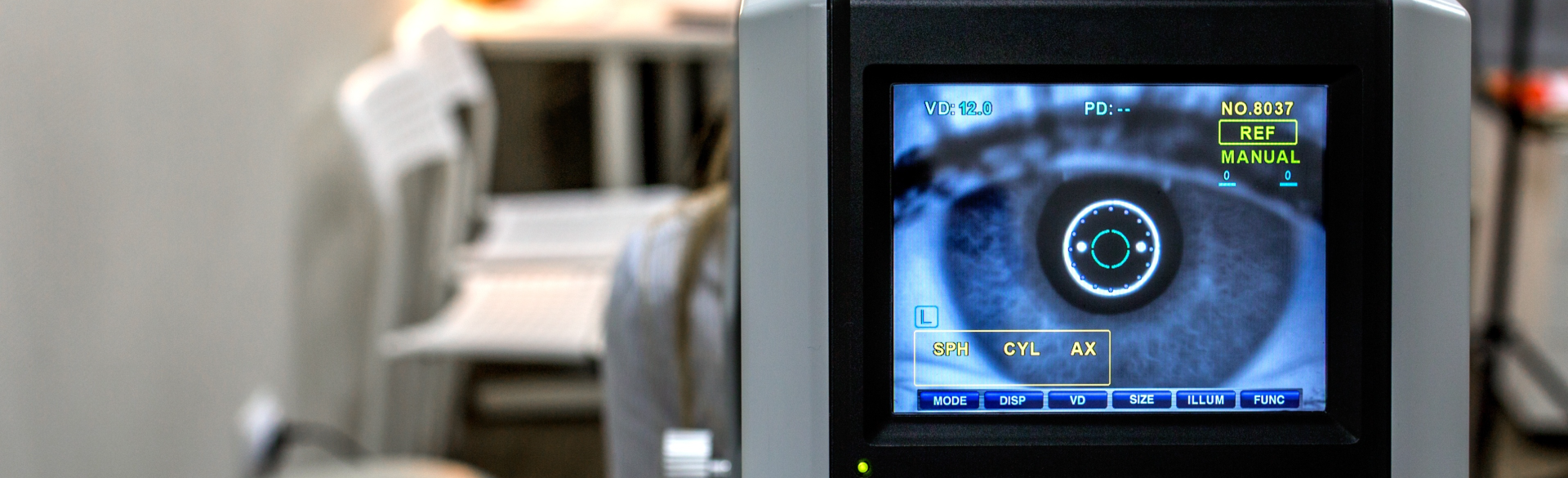
Glaucoma is a group of eye diseases that result in damage to the optic nerve; typically, this damage is caused by high pressure in the fluid inside the high (intraocular pressure, IOP). As it progresses, glaucoma can lead to loss of vision and eventually to permanent blindness. With over 2.2 million sufferers in the U.S. (and more than 60 million worldwide) glaucoma is the second leading cause of irreversible blindness.
Glaucoma treatment aims to reduce intraocular pressure using topical medications, laser treatments and surgery; for patients with advanced disease, filtering surgeries (aqueous shunting or trabeculectomy) are often used to help the eye’s inner fluid (the aqueous humor) drain, preventing loss of vision. While valuable, these surgical techniques are largely all-or-nothing, providing few ways for surgeons to customize the drainage rate; by five years post-surgery, they have a 50% failure rate.
 A research group led by Jeffrey Olson, MD, associate professor of ophthalmology, School of Medicine and member of the Rocky Mountain Lions Eye Institute, has developed an implantable device to give surgeons better control over the rate of drainage in glaucoma filtering surgeries. Importantly, the drainage rate can be adjusted not just at the time of the initial surgery, but months to years after surgery, decreasing surgical complications and improving patient outcomes.
A research group led by Jeffrey Olson, MD, associate professor of ophthalmology, School of Medicine and member of the Rocky Mountain Lions Eye Institute, has developed an implantable device to give surgeons better control over the rate of drainage in glaucoma filtering surgeries. Importantly, the drainage rate can be adjusted not just at the time of the initial surgery, but months to years after surgery, decreasing surgical complications and improving patient outcomes.
With early studies already successfully completed, Galaxy is applying for a Colorado Bioscience Discovery & Evaluation Grant (BDEG) to fund the first animal studies of the device at the CU Anschutz Medical Campus. In parallel with this study, Galaxy is seeking a licensing or co-development agreement to help bring the device to the market.
“The University is very excited about this agreement, and believes this partnership with Galaxy has great potential to benefit a patient population that needs alternative therapies beyond what’s currently available,” said David Poticha of the CU Technology Transfer Office.
The CU Technology Transfer Office pursues, protects, packages, and licenses to business the intellectual property generated from research at CU. The TTO provides assistance to faculty, staff, and students, as well as to businesses looking to license or invest in CU technology. See more at www.cu.edu/techtransfer.
Galaxy Ophthalmics is a Colorado-based corporation seeking to develop a number of innovative medical devices for ophthalmic therapies and surgeries leveraging the wealth of ophthalmic and general medical device development talent available not only at CU but across the Front Range.
###
Contact: Lindsay.Lennox@cu.edu, 303-735-5518