Thirteen-year-old Grace Hoyt received potentially the best birthday gift ever this month when pediatric ophthalmologists at the University of Colorado School of Medicine and Children’s Hospital Colorado administered the first treatment designed specifically to slow her vision loss associated with posterior column ataxia with retinitis pigmentosa (PCARP), a rare genetic condition that affects vision and the nervous system.
“It’s so incredible that she has this opportunity,” Susan Hoyt says of her daughter, who received the first treatment Aug. 24. “We’ve known that Grace is going to go blind, but to have the chance to stop the inexorable march forward is encouraging.”
The new treatment given with permission from the U.S. Food and Drug Administration as an investigational new drug is the first start-to-finish personalized medication given at Children’s Colorado developed in partnership with the CU Department of Ophthalmology.
“This is my first time applying for Investigational New Drug (IND) approval and the first new drug that I’ve given a patient,” says Emily McCourt, MD, associate professor and the Ponzio Family Chair for Pediatric Ophthalmology, who worked with researchers and physicians in the department, the newly-created CU Anschutz Investigational New Drug and Device (IND/IDE) Office, Boston Children’s Hospital, and non-profit drug developer n-Lorem to create the injectable medication that aims to slow down the patient’s vision loss.
“I learned through the process that sometimes you have to phone a friend, and thankfully we have a lot of expertise on the CU Anschutz campus,” she continues. “The IND office was so helpful, as was the research institute at Children’s Colorado.”
A mystery until diagnosis
McCourt met Grace 10 years ago when she was experiencing some motor delays, trouble seeing at night, and falling without responding in pain. One night, Grace put a sippy cup down and couldn’t find it at all.
“She was about a year-and-a-half old when we realized she couldn’t feel pain,” Susan says. “Then, about a year later, we were sitting her in her room one night and she couldn’t find her cup, but it was right in front of her.”
Soon after, McCourt first saw Grace and noticed that she had retinitis pigmentosa (RP), a blinding condition of the retina that develops over time. It is rare to experience as a young child, McCourt says. Typically people who have the disease are diagnosed in their 20s.
“At that time, we knew it was a clue to an overall diagnosis, but it was still difficult to put it all together,” she says. “Meanwhile, Grace was experiencing infections in the fingers along with broken bones that didn’t seem to hurt at all.”
Genetic testing revealed a few mutations, but none that McCourt says really made any sense. “A lot of the time you need two mutations to have a disease, and this patient only had one mutation in any gene that could explain her symptoms.”
The discovery of a second gene mutation, which researchers describe as a deep intronic mutation, only discoverable through whole genome sequencing, led to the PCARP diagnosis.
“It’s a really difficult disease,” says Marc Mathias, MD, associate professor of ophthalmology and retina specialist at the Sue Anschutz-Rodgers Eye Center, who assisted McCourt through the process of developing the treatment. “It doesn’t just affect the eyes and vision. There’s a systemic component as well, and the disease can develop slowly in childhood so it might not be recognized early on.”
In the last 50 years, PCARP has only been described in about 20 cases.
One patient, one treatment
Because Grace’s disease has a deep intronic mutation, her form of PCARP is amenable to a certain kind of therapy, called antisense oligonucleotide therapy (ASO), that attaches to RNA.
“Basically, you take these little molecules and put them together and make a drug that binds to the patient’s RNA to help the patient’s body make more normal protein and less abnormal protein,” McCourt explains. “We thought perhaps this therapy could work to slow down or stop the progression of the retinal disease.”
Austin Larson, a CU and Children’s Colorado geneticist who had recently given an ASO drug to another patient contacted a lab at Boston Children’s Hospital to see what they thought of the possibility of using an ASO therapy for Grace’s retina. The new partners got to work, using cells grown from Grace’s skin biopsy, which showed promising preliminary results.
From there, developing the treatment required testing, making sure the treatment would be safe for Grace, and finally getting the IND application approved by the FDA.
“Safety was goal number one,” Mathias says. “We wanted to do the right thing for the patient and design a study in a thoughtful way that protects her in the end.”
With the drug designed to treat one patient only, researchers had no control group, making it more challenging to show that the drug will be helpful over time, but studying the effectiveness of the therapy was similar to how scientists research drugs that reach bigger populations, too.
“It builds on a lot of experience we have with other clinical trials, and we went through a similar process,” Mathias says.
n-Lorem, a non-profit foundation dedicated to developing ASO technology to treat nano-rare patients, conducted testing for the trial. Once researchers were satisfied with the therapy, it was off to the FDA to submit an IND application.
“This whole process highlights the fact that medicine is a team sport,” McCourt says.
The power of asking 'what if'
Grace is expected to need the medication, through injections into the eyes, for the rest of her life.
“Because she doesn't feel pain, it's extremely important that Grace hangs on to whatever vision she has left so she can see her body, and evaluate herself for injuries” Susan says. “I am so grateful to Dr. McCourt for asking ‘what if’ and making the impossible happen for my girl.”
CU ophthalmologists will monitor her progress and watch for vision loss. McCourt and Mathias expect an injection may be necessary every three to six months. The treatment is being manufactured by n-Lorem for the rest of Grace’s life at no cost.
“It’s really taken a village to accomplish this,” echoes Mathias. “There has been support from many players including Grace, who has shown us all the reasons her family calls her Amazing Gracie.”
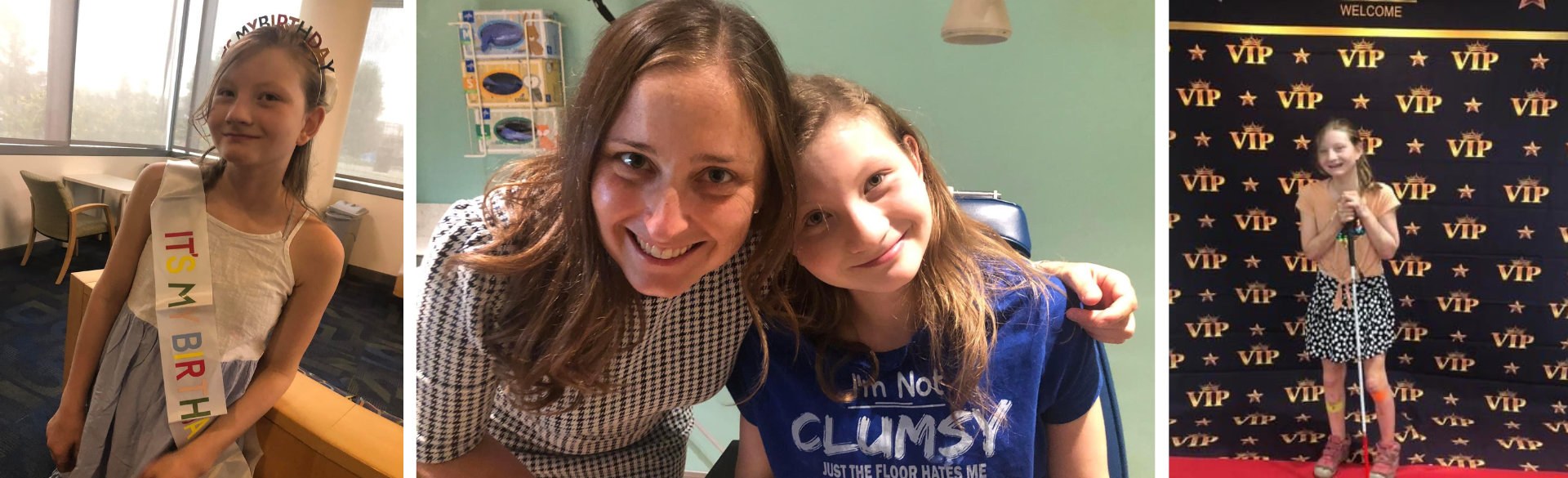



.png)
.png)