In a future perhaps not too far away, artificial intelligence and its subfield of machine learning (ML) tools and models, could help orthopedic physicians predict when a patient might encounter a bone fracture and what type.
“Right now, these kinds of tools aren’t widely used in clinical practice, but they are being used in research,” says Michael A. David, PhD, instructor of orthopedics at the University of Colorado Anschutz School of Medicine. “The appeal is that these tools can take a large amount of data and efficiently make sense of it with minimal human input.”
The use of machine learning — a broad computational field that “teaches” computers to automate tasks and find unexplored data patterns — is expected to accelerate those efforts, and David’s work is helping to push it forward.
His research integrates ML with spatial histopathology, transcriptomics, and medical imaging to study and diagnose post-traumatic and idiopathic joint conditions such as contracture, osteoarthritis, osteoporosis, and tendon ruptures. Oftentimes, this work entails a deep familiarity with the medical knowledge that’s required in orthopedics and the technological expertise that allows him to build and operate ML tools.
In a new review article published in Bone Reports, David explains that “machine learning might enable agnostic, automated analysis that accelerates discovery and improves clinical bone diagnosis and treatment monitoring.”
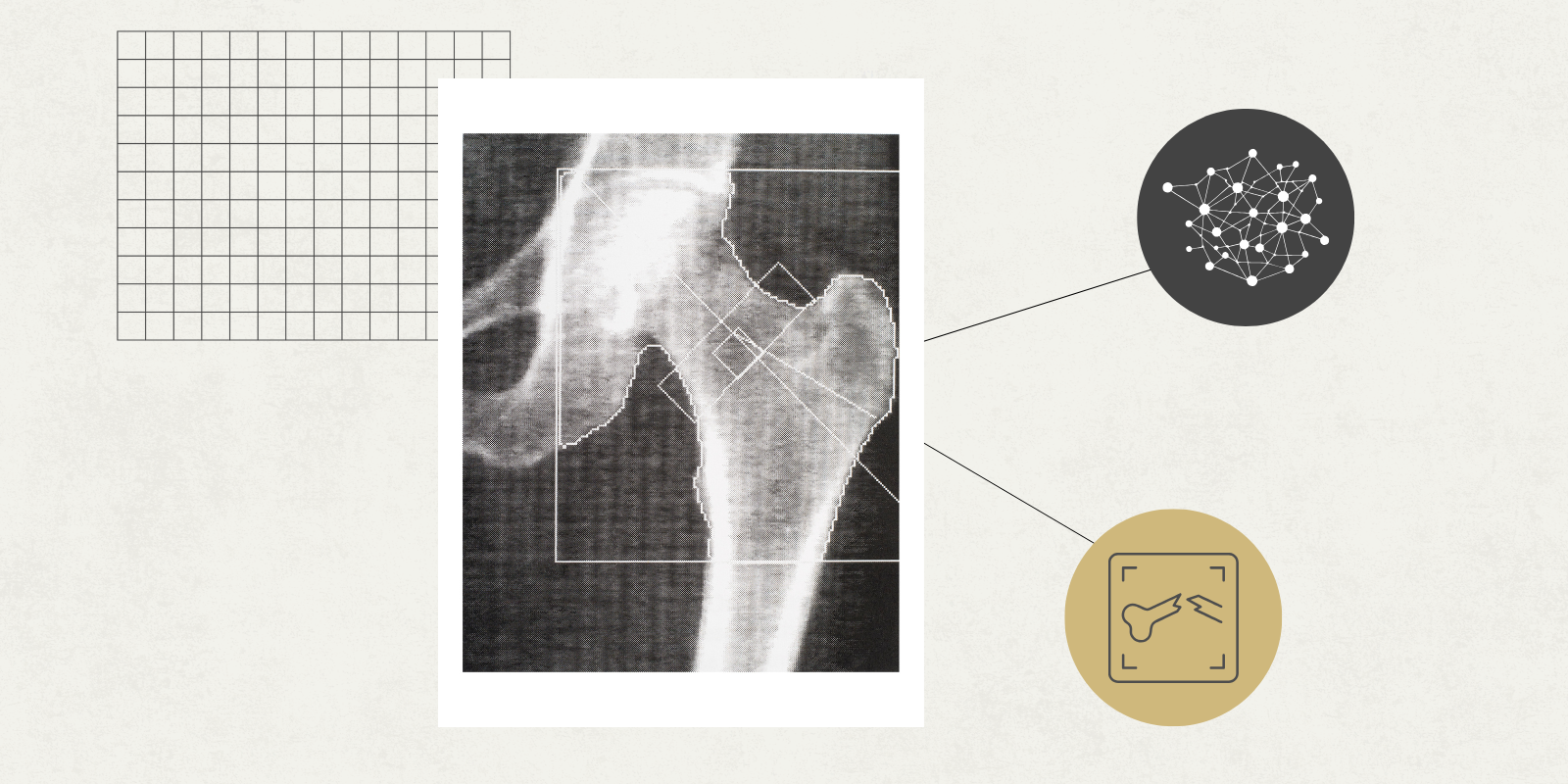
Utilizing bone imaging data with machine learning
One useful application of ML, among many, is that it can perform segmentation — dividing digital bone images into distinct bone types much faster than a human can. In orthopedic research, where bone imaging captures important data and large quantities of that data is needed to be effective, that’s a big perk.
“Segmenting bone data is the most laborious, time-consuming task,” says David, who has performed this work himself as a graduate student. “That's one area where ML excels in terms of accelerating the pace of discovery. It automates the segmentation and extracts quantitative data from the images.”
However, there are caveats. Not every bone or image contains the same information or is relevant to every research endeavor. Human expertise and analysis is still required for ML to be effective.
David’s review paper focuses on how researchers are using ML in two imaging techniques common in assessing bone structure: micro-computed tomography (microCT) and high resolution peripheral quantitative computed tomography (HRpQCT).
In these areas, ML “has been used primarily to enhance image resolution, automate bone segmentation, reduce the number of experimental variables, cluster data, and combine these with the prediction and classification of bone health and fracture risk,” David says.
While these breakthroughs of technology are impressive and meaningful, there’s still little chance that ML in orthopedics will replace radiologists. Instead, David says, these methods will appear in the form of clinical tools that will help health care providers make the best decisions for their patients.
Using data to cluster groups of patients will also benefit patients by learning more about subtypes or stages of diseases, such as osteoporosis.
“With ML, you can combine many types of data, like bone imaging, genetics, and even sleep cycle information, to see whether subgroups form. In terms of progress, I think we will see a lot coming from that space,” David says.
Preparing the next wave of scientists
In the coming decades, uses of ML will likely become more prominent, but to be helpful, researchers should deeply understand them.
“Although significant information is available in the public domain, particularly with the rise of large language ML models such as Deepseek, ChatGPT, Claude, and Gemini, the sheer breadth of tools, models, metrics, and interpretability measures creates a steep learning curve,” David says. “The time-consuming, arduous learning process can be a significant barrier to entry for bone researchers seeking to explore and adopt ML approaches.”
To lessen that barrier, David wrote the review paper and in the process created reproducible code for basic ML coding. He also developed SciNetX, a scientometric and bibliometric analytical pipeline and visualization software.
“SciNetX was born from the fact that when you complete a review study, you have to come up with a structure and show how all of the research papers you’ve looked at fit together,” he says.
By inputting various types of information, like the subject of the paper, keywords, authors, and more, the software creates visual networks, showing how different topics or investigators are related.
“It’s basically a map of the field,” David says. “As a new researcher in this area, I could look at this paper and see where this research is originating, who is doing it, and who they are collaborating with.”
David developed the software for working in orthopedics, but it could be applied almost anywhere, from various medical research efforts to other endeavors completely outside of medicine and scientific research. He’s currently working with CU Innovations to license the program so others can make use of it, too.
Ultimately, David’s envisions a symbiosis of human and computer interaction, and his goal is lower the barrier for people desiring to incorporate ML in digital bone imaging and uncover the vast networks of scientific knowledge.
“The whole idea is to invite more people into this world,” David says. “As I was developing SciNetX, I realized how powerful it could be. I want people coming to this field of research ready to learn, and this helps accomplish that.”