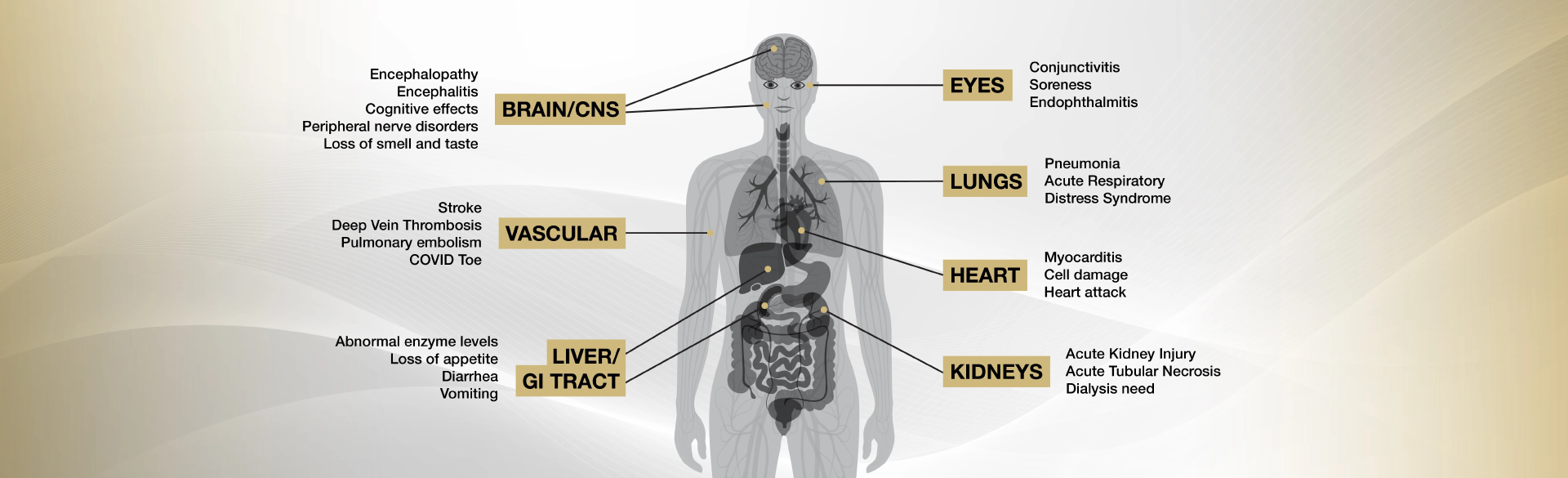
During the months-long pandemic, healthcare providers have seen a lot of things, often on levels they have never seen before. From brain fog and loss of smell to leg clots and purple toes, what began as a mysterious pulmonary disease has shown the world that its destructive powers far transcend the lungs.
While respiratory failure remains the most serious outcome of Coronavirus Disease 2019 (COVID-19), scientists have found evidence that SARS-CoV-2 can make its way into most major systems of the body using its spike proteins to attach and invade through ACE2 (angiotensin converting enzyme 2) receptors.
Below, experts from the School of Medicine at the University of Colorado Anschutz Medical Campus, share some of the effects they’ve seen on the body, from head to toe. But they note that the information must be kept in perspective.
The limited data focus almost exclusively on the minority of infected patients who end up hospitalized with COVID-19. And with the SARS-CoV-2 virus infecting upwards of 100 million (as of Jan. 26) people worldwide since its discovery a year ago, definitive connections are sparse. The need for many randomized studies remains.