A new study published in the Journal of the National Cancer Institute brings researchers and oncologists one step closer to better understanding the complexities of PD-1 inhibitors, a common type of immunotherapy, and their intracellular signaling on cancer behavior.
Until recently, cancer intrinsic PD-L1 signaling has been only partially reported in a descriptive manner. This new paper aims to provide a bigger picture of the effect PD-L1on cancer cells beyond the role of PD-L1 as a PD-1 ligand.
“We’re offering a more in-depth view of the cancer and immune cell interaction. This is important because by better understanding immunotherapy-driven signaling, we can better treat patients who haven’t had success with immunotherapy. Our findings show we can potentially achieve tumor control and provide patients with more successful cancer treatments,” said author Antonio Jimeno, MD, PhD, director of the Head and Neck Cancer Program at the University of Colorado School of Medicine and co-leader of Development Therapeutics Program at the University of Colorado Cancer Center. Both are located on the University of Colorado Anschutz Medical Campus.
Jimeno’s research is focused on achieving durable tumor control for head and neck squamous cell cancer (HNSCC) but he says these findings can be applicable for treating other cancers as well, since the interaction of PD-1 and PD-L1 is possibly the most validated immune target in human cancers.
More effective treatments for cancer patients
“Better understanding what makes cancer cells vulnerable to PD-L1 inhibition will allow oncologists to propose more effective treatments to patients with a range of cancers. This is particularly important for patients who aren’t currently benefiting from immunotherapy or they experience a delayed resistance,” Jimeno adds.
Currently, PD-L1 treatment is effective for less than 20% of HNSCC patients.
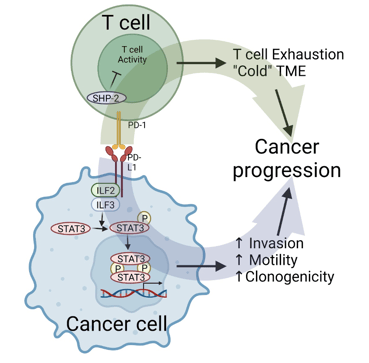 Jimeno’s and his team’s research found that upon binding to PD-1, PD-L1 triggers signaling that intrinsically alters the cancer cell phenotype, in addition to impacting the immune milieu. They found PD-L1 does this by direct interaction of its cytoplasmic domain with a group of intracellular proteins (the PD-L1 interactome) that were characterized in detail using a proximity labeling approach in live cells from several patients. PD-1 and PD-L1 interaction led to increased STAT3 signaling resulting in higher cancer cell clonogenicity, motility and invasion, as well as resistance to radiation and chemotherapy. Then the in vitro results were validated using a pharmacological inhibitor of STAT3, the main PD-L1 interactome partner, in humanized mice bearing tumors donated by Coloradans with head and neck cancer.
Jimeno’s and his team’s research found that upon binding to PD-1, PD-L1 triggers signaling that intrinsically alters the cancer cell phenotype, in addition to impacting the immune milieu. They found PD-L1 does this by direct interaction of its cytoplasmic domain with a group of intracellular proteins (the PD-L1 interactome) that were characterized in detail using a proximity labeling approach in live cells from several patients. PD-1 and PD-L1 interaction led to increased STAT3 signaling resulting in higher cancer cell clonogenicity, motility and invasion, as well as resistance to radiation and chemotherapy. Then the in vitro results were validated using a pharmacological inhibitor of STAT3, the main PD-L1 interactome partner, in humanized mice bearing tumors donated by Coloradans with head and neck cancer.
This research brings doctors and researchers one step closer to understanding the complexities of PD-L1/PD-1 and the relevance it has for targeting most solid tumors and stopping the progression of cancer.
“The dissection of PD-L1 cancer and immune cell interaction is much more coordinated and has wider and deeper implications than we thought. This is an evolutionary efficient and elegant mechanism for the cancer cell to trigger invasion once immune evasion has achieved. It shows that we need to research immunotherapy with sufficiently complex viewpoints and tools to actually know what’s going on,” Jimeno says. “This will help us understand the mechanistic events of PD-L1, intrinsically and extrinsically to the cancer cell. It’s a vital area of focus for cancer biology and immunology.”




