A sweeping international study of genetic data from nearly 2.9 million people has uncovered hundreds of previously-unknown genetic variants linked to thyroid cancer and other thyroid diseases, offering a potential roadmap for earlier cancer detection and a better way to predict which patients are at higher risk of developing more dangerous thyroid cancers.
The study marks a big step toward what the researchers call "genetically informed" thyroid care, in which a patient's DNA pattern could help guide care from diagnosis through treatment.
University of Colorado Anschutz Cancer Center members Nikita Pozdeyev, MD, and Christopher Gignoux, PhD, are corresponding authors of the study, published last month in the journal Nature Genetics. Pozdeyev, an endocrinologist and thyroid cancer specialist, is an assistant professor in the CU Anschutz Department of Biomedical Informatics. His research focuses on using “big data” to inform clinical care. Gignoux is a statistical geneticist and a professor of biomedical informatics.
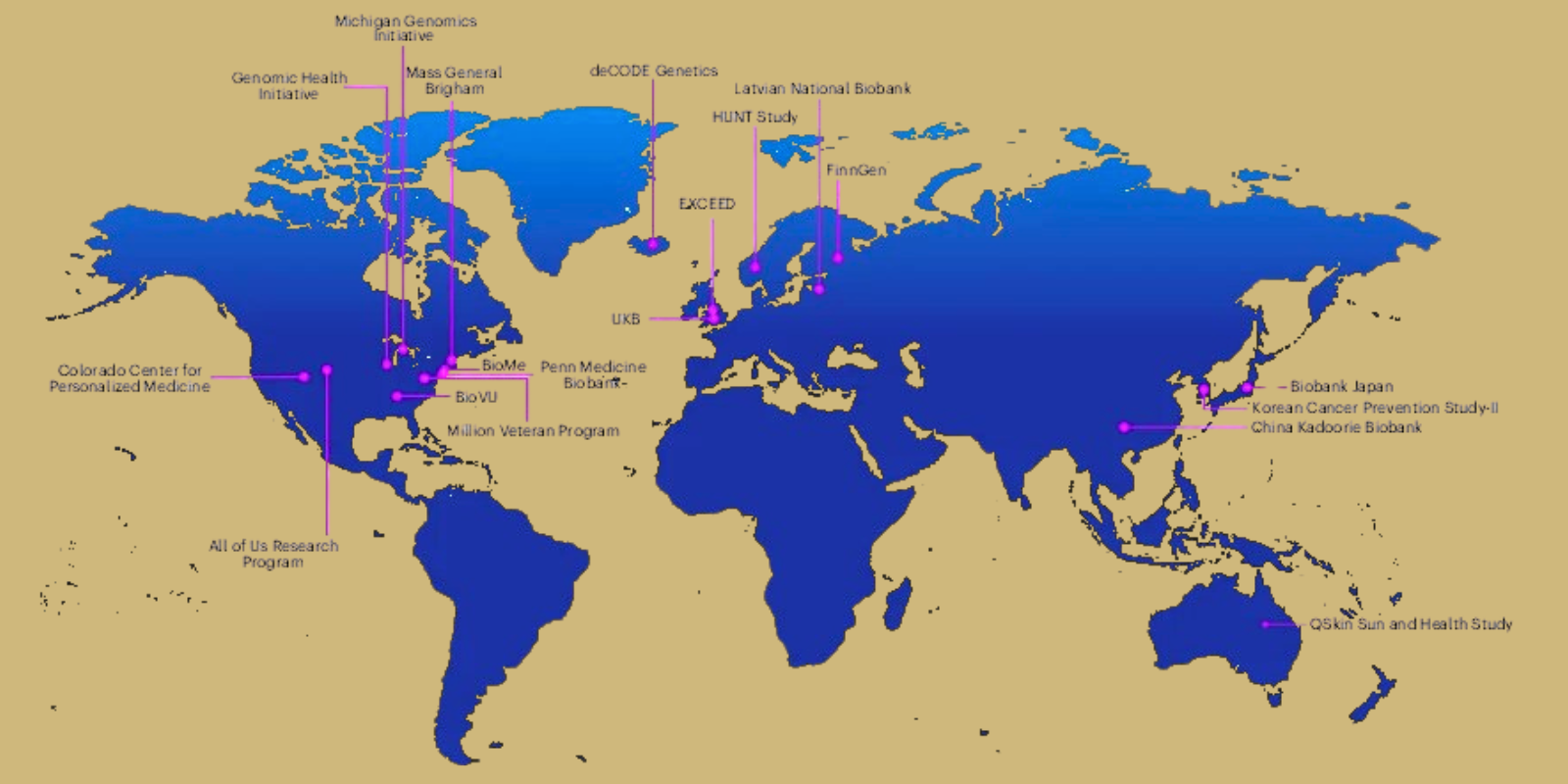
The study is the largest genetic analysis of thyroid disease ever conducted, Pozdeyev says. It involved 19 research biobanks across 10 countries and four continents that are part of the global Virtual Thyroid Biopsy Consortium.
The consortium includes the biobank at the Colorado Center for Personalized Medicine (CCPM) at CU Anschutz, one of the largest academic biospecimen repositories in the United States, with over 100,000 blood and DNA samples from volunteer participants. CCPM is the only institution in the Rocky Mountains in the group.
“In statistical genetics, no single biobank or single university, or even a single country, has enough data to generate useful insights from it,” Pozdeyev says. “In genetics, more data is always better. This way, we work with the world.”
→ Does Using GLP-1 Drugs, Like Zepbound or Wegovy, Increase Your Risk of Thyroid Cancer?
About thyroid cancer
The thyroid — a small, butterfly-shaped gland in the neck — regulates metabolism, heart rate, and many other bodily functions. According to the American Thyroid Association, more than 12% of Americans will develop some sort of thyroid condition during their lifetime, yet up to 60% of those with thyroid disease are unaware of their condition.
The American Cancer Society projects about 45,240 new cases of thyroid cancer in the United States this year, including about 840 in Colorado, and about 2,320 deaths nationwide. The cancer is almost three times more common in women than in men.
Thyroid cancer is often diagnosed at a younger age than most adult cancers. The average age at diagnosis is 51. While many cases are slow-growing and often successfully treated, some are aggressive and deadly.
Non-cancerous autoimmune thyroid conditions like Graves' disease (which causes an overactive thyroid) and Hashimoto's thyroiditis (which typically leads to an underactive thyroid) affect millions and can lead to elevated cardiovascular risks.
→ AI Can Recognize Thyroid Nodules That Are Very Unlikely To Be Cancerous
A clinical need
Pozdeyev says that as a clinician, most of his research projects “start with a clinical need. It would be helpful to be able to estimate which individuals are at risk of developing thyroid cancer, and especially aggressive thyroid cancer.”
He says thyroid cancer “has a very high heritability, which means that a great deal of risk is defined by what you inherit from your parents. A lot of it is genetics. We realized we didn’t have a good way of quantifying that risk, and we didn’t even know the genes and variants that contribute to that risk. That’s why we started this study, to discover those.”
Samantha White, a senior data scientist in Pozdeyev’s lab, in collaboration with the rest of the team, combined data for millions of sites in the genome to identify variants associated with five thyroid conditions: thyroid cancer, benign nodular goiter, Graves' disease, lymphocytic thyroiditis, and primary hypothyroidism. The team identified 883 genetic associations with thyroid disease, 570 of which had never been found before.
The study found distinct genetic fingerprints that help to explain why some patients develop benign growths called thyroid nodules while others develop cancer.
Many genes identified in the study were associated with both benign and malignant thyroid nodules, suggesting they predispose people to developing both. But a set of genes responsible for regulating the cell cycle, repairing DNA damage, and responding to DNA breaks was linked predominantly to thyroid cancer — not benign disease.
→ Department of Defense Award Supports CU Thyroid Cancer Research
Polygenic risk score
Following up on earlier work by Pozdeyev and several CU Anschutz colleagues, the new study found that a polygenic risk score — a numerical summary of a person's inherited cancer risk based on their DNA — could improve classification of thyroid nodules as benign or cancerous. People in the top 10% of the risk score were found to be nearly eight times more likely to have a malignant nodule than those in the bottom 10%.
Each year in the United States, more than 600,000 fine-needle aspiration biopsies are performed to determine whether a thyroid nodule is cancerous. The vast majority — about 92% — come back benign, indeterminate, or inconclusive. The study authors envision the polygenic risk score being used alongside existing ultrasound-based risk tools to reduce unnecessary biopsies, or to flag cases that might otherwise be missed.
The study also found an association between the polygenic risk score and cancer aggressiveness. Higher risk scores were linked to larger tumors, cancer spreading to multiple sites within the thyroid, lymph node metastases, and a dangerous pattern called extranodal extension, where cancer breaks through the wall of a lymph node and invades surrounding tissue. Higher scores were also associated with the risk of disease recurrence after treatment.
“In thyroid cancer, most patients are cured with surgery and radiation,” Pozdeyev says. “But a small subset will develop an incurable disease and die from it. Our hope is that, with this risk score, we can better identify those people and diagnose them early, before they get into trouble.”
→ CU Research Uncovers New Driver of Thyroid Cancer Metastasis
A hobby that’s a job
Pozdeyev cautions that the risk score will need calibration and testing in clinical trials before entering routine practice. He and CU Anschutz Cancer Center member Bryan Haugen, MD, professor of medicine in the Division of Endocrinology, Metabolism and Diabetes, are designing a clinical trial now and seeking funding.
Pozdeyev also notes that the majority of participants in the dataset used for the study were of European and East Asian ancestry, limiting the study's scope in detecting associations specific to people of African or Latin American backgrounds. As the consortium grows, the team plans to expand its ancestry-specific analyses to ensure findings are relevant across populations, he says.
Nevertheless, Pozdeyev hopes the study helps usher in a future where patients with thyroid nodules get genetic screening to help determine not just whether they have cancer, but how dangerous it is likely to be.
Pozdeyev says he’s thrilled to be able to wed his interests in endocrinology and informatics in his research unlocking cancer’s secrets. “I've been programming and writing code for fun since I was a kid,” he says with a grin, “and now I do my hobby as a job.”
Map at top shows the location of the biobanks participating in the global Virtual Thyroid Biopsy Consortium, including the Colorado Center for Personalized Medicine at CU Anschutz.